Chapter 10.1: Storage Lipids
-
Upload
cadman-mckinney -
Category
Documents
-
view
66 -
download
0
description
Transcript of Chapter 10.1: Storage Lipids

Chapter 10.1: Storage Lipids
CHEM 7784
Biochemistry
Professor Bensley

CHAPTER 10.1 Storage Lipids
– Biological roles of lipids – Structure and properties of storage lipids
Chapter Objectives: To understand the

Lipids: Structurally Diverse Class
• Low solubility in water• Good solubility in nonpolar solvents




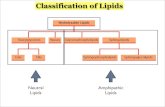
Classification of Lipids
Based on the structure and function:
1. Lipids that contain fatty acids (complex lipids):Storage lipids and membrane lipids
2. Lipids that do not contain fatty acids: Cholesterol, terpenes, …

Lipid Subclasses


Fatty Acids
• Carboxylic acids with hydrocarbon chains containing from 4
to 36 carbons
• Almost all natural fatty acids have an even number of
carbons
• Most natural fatty acids are unbranched
• Saturated:
• Monounsaturated:
• Polyunsaturated:


Common Saturated Fatty Acids
common name IUPAC name melting point (Co)
12:0 laurate dodeconoate 44
14:0 myristate tetradeconoate 52
16:0 palmitate hexadeconoate 63
18:0 stearate octadeconoate 70
20:0 arachidate eicosanoate 75
22:0 behenate docosanoate 81
24:0 lignocerate tetracosanate 84

Common Unsaturated Fatty Acids
common name IUPAC namemelting point
(Co)
16:0 palmitate hexadeconoate 63
16:1 9 palmitoleate cis-9-hexadeconoate -0.5
18:0 stearate octadeconoate 70
18:1 9 oleate cis-9- octadeconoate 13
18:2 9,12 linoleate cis-9,12- octadeconoate -9
18:3 9,12,15 linolenate cis-9,12,15- octadeconoate -17
20:0 arachidate eicosanoate 75
20:4 5,8,11,14 arachindonate cis- 5,8,11,14-eicosatetraenoate -49

Physical Properties and Conformation of Saturated Fatty Acids
• Solubility decreases as the chain length increases
• Melting point increases as the chain length increases
• The saturated chain tends to adopt extended conformations
• The double bonds in natural unsaturated fatty acids are commonly in cis configuration
• This introduces a kink in the chain


Melting Point and Double Bonds
• Saturated fatty acids pack in a fairly orderly way– extensive favorable interactions
• Unsaturated cis fatty acid pack less regular due to the kink– Less extensive favorable interactions
• It takes less thermal energy to disrupt disordered packing of unsaturated fatty acids:– unsaturated cis fatty acids have a lower melting point

Melting Points of
Fatty Acids
18:0 18:1 18:3
70o C 13o C -17o C


Trans Fatty Acids
• Trans fatty acids form by partial dehydrogenation of unsaturated fatty acids
• A trans double bond allows a given fatty acid to adopt an extended conformation
• Trans fatty acids can pack more regularly, and show higher melting points than cis forms


Triacylglycerols (fats and oils)
• Majority of fatty acids in biological systems are found in the form of triacylglycerols
• Triacylglycerols are the primary storage form of lipids (body fat)
• Triacylglycerols are less soluble in water than fatty acids due to the lack of charged carboxylate group
• Triacylglycerols are less dense than water: fats and oils float



Waxes
• Waxes are esters of long-chain saturated and unsaturated fatty acids with long-chain alcohols
• Insoluble and have high melting points
• Beeswax is a mixture of a large number of lipids, including esters of triacontanol, and a long-chain alkane hentiacontane