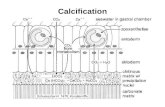
Calcification - growth of the reef. In ocean, mostly find 3 forms of CaC0 3 Calcite –Mostly of...
-
Upload
luke-mitchell -
Category
Documents
-
view
221 -
download
0
Transcript of Calcification - growth of the reef. In ocean, mostly find 3 forms of CaC0 3 Calcite –Mostly of...

Calcification - growth of the reef

• In ocean, mostly find 3 forms of CaC03
• Calcite– Mostly of mineral origin
• Aragonite– Fibrous, crystalline form, mostly from corals
• Magnesian calcite– Smaller crystals, mostly plant origin

Calcification
Calcite
Aragonite
Magnesian calcite (Mg carbonate)

• Examples:
organism CaCO3
Molluscs calcite & aragonite
Corals just aragonite
Some green algae just aragonite
Red algae magnesian calcite
Sponges aragonite (with silica)
Some bryozoans all 3

Corals
• remove Ca++ & CO3-- from seawater
• Combines them to CaCO3
• transports them to base of polyp
– Calcicoblastic epidermis
• minute crystals secreted from base of polyp
• Energy expensive– Energy from metabolism of algal PS products

Calcification

CO2 and seawater
• What forms of C are available to the coral ?
• Organic and inorganic forms
• DIC - dissolved inorganic carbon– CO2 (aq)
– HCO3-
– CO3--

• DIC comes from:
– Weathering– dissolution of oceanic rock– Run-off from land– Animal respiration– Atmosphere– etc.

• DIC in ocean constant over long periods
• Can change suddenly on local scale– E.g. environmental change, pollution
• Average seawater DIC = 1800-2300 mol/Kg
• Average seawater pH = 8.0 - 8.2
• pH affects nature of DIC

Carbon and Seawater
• normal seawater - more HCO3- than CO3
--
• when atmospheric CO2 dissolves in water
– only 1% stays as CO2
– rest dissociates to give HCO3- and CO3
--

H2O + CO2 (aq) H2CO3 HCO3- + H+ (1)
HCO3- CO3
-- + H+ (2)
equilibrium will depend heavily on [H+] = pH
relative amounts of different ions will depend on pH


dissolved carbonate removed by corals to make aragonite
Ca++ + CO3--
CaCO3 (3)
pulls equilibrium (2) over, more HCO3- dissociates to CO3
--
HCO3- CO3
-- + H+ (2)
removes HCO3-, pulls equilibrium in eq (1) to the right
H2O + CO2 (aq) H2CO3 HCO3- + H+ (1)
more CO2 reacts with water to replace HCO3-, thus more CO2 has to
dissolve in the seawater

Can re-write this carbon relationship:
2 HCO3- CO2 + CO3
-- + H2O
• used to be thought that
– symbiotic zooxanthellae remove CO2 for PS
– pulls equation to right
– makes more CO3-- available for CaCO3 production by polyp
• No

• demonstrated by experiments with DCMU – stops PS electron transport, not CO2 uptake
• removed stimulatory effect of light on polyp CaCO3 deposition
• therefore, CO2 removal was not playing a role
• also, in deep water stony corals– if more food provided, more CaCO3 was deposited
– more energy available for carbonate uptake & CaCO3 deposition

• Now clear that algae provide ATP (via CHO) to
allow polyp to secrete the CaCO3 and its
organic fibrous matrix
• Calcification occurs 14 times faster in open than
in shaded corals
• Cloudy days: calcification rate is 50% of rate on
sunny days
• There is a background, non-algal-dependent rate

Environmental Effects of Calcification
• When atmospheric [CO2] increases, what happens to calcification rate ?
– goes down
– more CO2 should help calcification ?
– No

• Add CO2 to water– quickly converted to carbonic acid
– dissociates to bicarbonate:
H2O + CO2 (aq) H2CO3 HCO3- + H+ (1)
HCO3- CO3
-- + H+ (2)
• Looks useful - OK if polyp in control, removing CO3--

• Add CO2 to water– quickly converted to carbonic acid
– dissociates to bicarbonate:
H2O + CO2 (aq) H2CO3 HCO3- + H+ (1)
HCO3- CO3
-- + H+ (2)
• Looks useful - OK if polyp in control, removing CO3--
• BUT, if CO2 increases, pushes eq (1) far to right

• Add CO2 to water– quickly converted to carbonic acid
– dissociates to bicarbonate:
H2O + CO2 (aq) H2CO3 HCO3- + H+ (1)
HCO3- CO3
-- + H+ (2)
• Looks useful - OK if polyp in control, removing CO3--
• BUT, if CO2 increases, pushes eq (1) far to right
• [H+] increases, carbonate converted to bicarbonate

• So, as more CO2 dissolves,
• more protons are released
• acidifies the water
• the carbonate combines with the protons
• produces bicarbonate
• decreases carbonate concentration


• Also, increase in [CO2]
– leads to a less stable reef structure– the dissolving of calcium carbonate
H2O + CO2 + CaCO3 2HCO3- + Ca++

• Also, increase in [CO2]
– leads to a less stable reef structure– the dissolving of calcium carbonate
H2O + CO2 + CaCO3 2HCO3- + Ca++
• addition of CO2 pushes equilibrium to right
– increases the dissolution of CaCO3

• anything we do to increase atmospheric [CO2] leads to various deleterious effects on the reef:
• Increases solubility of CaCO3
• Decreases [CO3--] decreasing calcification
• Increases temperature, leads to increased
bleaching
• Increases UV - DNA, PS pigments etc.

Reef Photosynthesis

Productivity
• the production of organic compounds from inorganic atmospheric or aquatic carbon sources – mostly CO2
• principally through photosynthesis– chemosynthesis much less important.
• All life on earth is directly or indirectly dependant on primary production.

gC/m2/d
TropicalCoral Reef 4.1 - 14.6
Tropical open ocean 0.06 - 0.27
Mangrove 2.46
Tropical Rain Forest 5.5
Oak Forest 3.6

Productivity
• no single major contributor to primary production on the reef
• a mixture of photosynthetic organisms– can be different at different locations

• net productivity values (varies with location):
gC/m2/d
Calcareous reds 1 - 6
Halimeda 2 -3
Seagrass 1 - 7
N.S. kelp 5

• Overall productivity of the reef:
4.1 - 14.6 gC/m2/d
• from– epilithic algae, on rock, sand etc., – few phytoplankton– seagrasses– Zooxanthellae (in coral etc.)– Fleshy and calcareous macroalgae

• One obvious differences between different algae is their colour
• Different colours due to the presence of different photosynthetic pigments

Light
ReflectedLight
Chloroplast
Absorbedlight
Granum
Transmittedlight

Light and Photosynthesis
• Air & water both absorb light– a plant at sea level receives 20% less light than
a plant on a mountain at 4,000m
– this reduction occurs faster in seawater – depends a lot on location
• get 20% light reduction in 2m of tropical seawater
• get 20% light reduction in 20cm of Maritime seawater

• a very specific part of the EM spectrum
• PAR
• Photosynthetically Active Radiation
• 350-700 nm

Gammarays X-rays UV Infrared
Micro-waves
Radiowaves
10–5 nm 10–3 nm 1 nm 103 nm 106 nm1 m
106 nm 103 m
380 450 500 550 600 650 700 750 nm
Visible light
Shorter wavelength
Higher energy
Longer wavelength
Lower energy

• Measure it as IRRADIANCE– moles of photons per unit area per unit time– mol.m-2.s-1
– E = Einstein = 1 mole of photons
E.m-2.s-1

• As light passes through seawater it gets ABSORBED & SCATTERED – = ATTENUATION (a reduction in irradiance)
• pure water– attenuation lowest at 465nm
– increases towards UV and IR ends of spectrum
• TRANSMITTANCE is highest at 465nm
• not dealing with pure water– Seawater has all kinds of dissolved salts, minerals,
suspended material etc.:


• Attenuation is different in different locations - different light transmittance spectra:
To fully exploit a particular location, marine plants have a wide variety of PS pigments they can use.

Chloroplast
Mesophyll
5 µm
Outermembrane
Intermembranespace
Innermembrane
Thylakoidspace
Thylakoid
GranumStroma
1 µm

CO2
CALVINCYCLE
O2
[CH2O](sugar)
NADP
ADP+ P i
An overview of photosynthesis
H2O
Light
LIGHT REACTIONS
Chloroplast
ATP
NADPH

Light Reactions
• In the thylakoid membrane,
– chlorophyll molecules, other small molecules & proteins, are organized into photosystems
– photosystems composed of a reaction center surrounded by a number of light-harvesting complexes (LHC)
• LHC = pigment molecules bound to proteins

• LHC = pigment molecules bound to proteins
• funnel energy of photons to the reaction center
• reaction-center chlorophyll absorbs energy– One of its electrons gets bumped up to a primary
electron acceptor– electron transport– ATP & NADPH production

Photosystems
Primary electionacceptor
Photon
Thylakoid
Light-harvestingcomplexes
Reactioncenter
Photosystem
STROMAT
hyla
koid
mem
bran
e
Transferof energy
Specialchlorophyll amolecules
Pigmentmolecules
THYLAKOID SPACE(INTERIOR OF THYLAKOID)
e–

Light• The visible light spectrum includes
– the colors of light we can see– the wavelengths that drive photosynthesis
• Photosymthetic pigments absorb light

Gammarays X-rays UV Infrared
Micro-waves
Radiowaves
10–5 nm 10–3 nm 1 nm 103 nm 106 nm1 m
106 nm 103 m
380 450 500 550 600 650 700 750 nm
Visible light
Shorter wavelength
Higher energy
Longer wavelength
Lower energy

Light
ReflectedLight
Chloroplast
Absorbedlight
Granum
Transmittedlight

• different pigments have different absorption spectra
• combine in different amounts in different species to give each a unique absorption spectrum
• tells us which wavelengths of light are being absorbed (and thus it’s colour)

Ab
sorp
tion
of
ligh
t b
ych
loro
pla
st p
igm
en
ts
Chlorophyll a
Wavelength of light (nm)
Chlorophyll b
Carotenoids
Absorption spectra of pigments

• doesn’t tell us what the alga is doing with the light
• For this you need to look at the ACTION SPECTRUM– measures photosynthesis at different
wavelengths

• The action spectrum of a pigment
– show relative effectiveness of different wavelengths of radiation in driving photosynthesis
• Plots rate of photosynthesis versus wavelength


Marine PS pigments
• 3 major groups of PS pigments in marine organisms
– Chlorophylls– Phycobiliproteins– Carotenoids

• Chlorophyll a is essential
– find it in all plants and algae
• the other pigments are accessory pigments
– in the antennae complexes – funnel electrons to chlorophyll a in the reaction
centres

• 5 types of chlorophyll commonly found in marine organisms
• all are tetrapyrrole rings with Mg++ in the middle
• chlorophyll a, b, c1, c2 & d
• a all green plants and algae• b Chlorophyceae• c1 & c2 Phaeophyceae• d Rhodophyceae

• Chlorophyll a– Is the main photosynthetic pigment
• Chlorophyll b– Is an accessory pigment
C
CH
CH2
CC
CC
C
CNNC
H3C
C
CC
C C
C
C
C
N
CC
C
C N
MgH
H3C
H
C CH2CH3
H
CH3C
HHCH2
CH2
CH2
H CH3
C O
O
O
O
O
CH3
CH3
CHO
in chlorophyll a
in chlorophyll b
Porphyrin ring:Light-absorbing“head” of moleculenote magnesiumatom at center
Hydrocarbon tail:interacts with hydrophobicregions of proteins insidethylakoid membranes ofchloroplasts: H atoms notshown
Accessory pigments absorb different wavelengths of light and pass the energy to chlorophyll a

• Also a wide range of carotenoids– C40 TETRATERPENES– very hydrophobic– sit in membranes
• 2 types of carotenoids
– CAROTENES (hydrocarbons)– XANTHOPHYLLS (have 1 or 2 oxygens)

-CAROTENE is the most common carotenoid in marine organisms
• often see a mixture of -CAROTENE & FUCOXANTHIN (another carotenoid) in the Phaeophyceae– gives the brown colour

• PHYCOBILINS are linear tetrapyrroles attached to proteins– red pigments
– no ring, no chelation of a metal
• Only found in Rhodophyceae & Cyanophyceae– and a few species of Cryptophyceae