Bladder Cancer Diagnostic-Initial Team Project
-
Upload
sagar-desai -
Category
Health & Medicine
-
view
336 -
download
2
description
Transcript of Bladder Cancer Diagnostic-Initial Team Project

ACDS Laboratories
A new biomarker for bladder cancer diagnosis
Mirza AhmedKaty Chun
Sagar DesaiNik Sanyal
August 29, 2013

Who We Are
Dedicated to the advancement of biomarker development to address unmet medical needs and improve patient health

What is Bladder Cancer?
Stages of bladder cancerhttp://www.urologyhealth.org/urology/articles/images/anatomy_bladdercancer.jpg
1. Reasonably large population- Fourth most common cancer in men and eighth most common cancer in women5
- 72,570 new cases in the US in 20132
2. Importance of early detection- 96.4% 5-year relative survival rate if diagnosed in stage 0 or 1
- 15,210 deaths in the US in 20132
3. Costly expenses- Highest lifetime treatment costs per patient of all cancers 3
- “The natural history of bladder cancer is one of recurrence and progression necessitating long-term evaluation and monitoring”5
2National Cancer Institute, Bladder Cancer3Sievert et al., World J Uro. 20095Gilbert et al., Urology. 2003

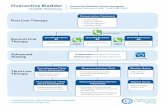
Current clinical flow for the diagnosis of bladder cancer

Sensitivity and specificity- Sensitivity (true positive rate) measures the proportion of actual positives that are correctly identified by the test4
- Specificity (true negative rate) measures the proportion of negatives that are correctly identified by the test4
4Altman & Bland, BMJ, 1994

Current diagnostic methods- Voided urine cytologyPresence of abnormal (cancerous) cells in urineIssues: low sensitivity (43.8%), results not available immediately 5
- CystoscopyEndoscopy via urethra Issues: Invasive, dependent on operator skill,inability to detect recurrences in the upper urinary tractrigid cystoscopy - painful and needs general anesthesia, may lead to scarring
flexible cystoscopy - does not allow removal of tissue5
Cystoscopy of the bladder http://www.mayoclinic.com/images/image_popup/
r7_cystoscopy.jpg
5Gilbert et al., Urology. 2003

Demand for a better biomarker
- Need exists for biomarker that is:- High sensitivity and specificity- Easily detected using a non-invasive procedure
- cost-effective- Several candidate biomarkers 6,7, but most have drawbacks
6Urquidi et al., Urology. 20127Liu et al., BMC Cancer. 2013

Selected Biomarkers1) miRNA - 135b/15b/1224-3p
2) DD23 antigen
3) Protein markers - VEGF, ApoE & IL-8

1. MicroRNAA combination of 3 miRNAs - 135b/15b/1224-3p - appear as promising stable biomarkers8
AssayUrinary samples (n = 121) from patients with bladder cancer and age-matched controls (n = 53) were analyzed using real-time PCR for changes in expression levels.
Clinical validitySensitivity: 94%Specificity: 51%Overall accuracy: 86%
8Miah et al., British Journal of Cancer. 2012

2. DD23 antigenAnti-DD23 antibody shows increased sensitivity of urinary based detection of UCC when combined with cytology5
AssayUrinary samples (n = 151) from patients with a history of bladder cancer were evaluated for DD23 antigen expression using alcohol-fixed urinary cytology
Clinical validity Sensitivity (%)
Specificity (%)
Cytology
43.8 92.0
Cytology + DD23
78.1 58.6
5Gilbert et al., Urology. 2003

3. Protein markers
A combination of 3 proteins - VEGF/ApoE/IL-8 - appear as a highly accurate biomarker10 - Vascular endothelial growth factor (VEGF)- Apolipoprotein E (ApoE)- Interleukin-8 (IL-8)
AssayVoided urine samples from individuals without previous history of UCC (n = 63) and individuals with newly diagnosed UCC (n=64) were quantified by Enzyme-Linked Immunosorbent Assay (ELISA)
Clinical validitySensitivity: 90%Specificity: 97%Overall accuracy: 93%
10 Goodison et al., PLOSone. 2012

Scientific assessmentElevated levels of biomarkers in bladder cancer individuals
10 Goodison et al., PLOSone. 2012

VEGF (Vascular Endothelial Growth Factor)Induces blood vessel formation (Angiogenesis) which helps sustains tumor growth.Frequently upregulated during bladder6, prostate12, breast cancer 13. Anti-cancer drug Bevacizumab (Avastin) targets VEGF receptor binding. ApoE (Apolipoprotein E)A type of lipoprotein found in chylomicron intermediate-density lipoprotein (IDL). In breast cancer, ApoE is upregulated via HIF1α14. IL-8 (Interleukin-8)Signaling factor produced by endothelial cells and macrophages. Promotes angiogenesis during hypoxia and environmental stress. Promotes cell proliferation in lung 15 and colon cancer16 .
Scientific assessment VEGF, ApoE and IL-8 in cancer
Waugh DJ, Wilson C. Clin Cancer Res. 2008 Nov 1;14(21)
6Urquidi et al., Urology. 201212Pratheeshkumar et al., PLoS One, 201213Marty and Pivot, Eur. J. Cancer, 200814Papi et al, PLoS One, 201315Luppi et al, Lung Cancer, 200716Ning et al, Int. J. Cancer, 2011

Scientific assessmentAdvantages of ELISA
- Rapid- Low detection limits - Low per sample cost- High-throughput (96 or 384-well format)
biology.arizona.edu/immunology/activities/elisa/graphics/elisa_plate.gif
Epitomics Inc.

Revised clinical flow for the diagnosis of bladder cancer

Patient ValueEarly, highly-accurate, non-invasive diagnosis
- Early stage diagnosis increases have long-term survival rates greater than 80%2
- Accurate diagnosis allows appropriate treatment and prevents unnecessary surgery- Avoid the need for invasive cystoscopy
Easy, long-term monitoring
- Cost-effective- Can be performed in outpatient setting
2National Cancer Institute, Bladder Cancer

Economic ValueEconomic analysis: Current prices
Cytology
Average cost = $100 17
Cystoscopy
Current Medicare reimbursement = $379 non-facility18
17Lotan and Roehrborn, J. Urology. 200218 CMS.gov, Centers for Medicare and Medicaid Services, Cytology

Current pricing for the diagnosis & monitoring of bladder cancer
$100 + $379 = $479
$100 + $379 = $479

Economic ValueEconomic analysis: Recurrences and monitoring
80% of bladder cancer patients survive 5-years19.
Long-term monitoring of bladder cancer patients:- Cytology and cystoscopy:
Every 3 months during first 2 years (8 procedures)Every 6 months during next 2 years (4 procedures)Every 12 months during subsequent years 20
1initial + 8 + 4 + 1 = 14 procedures/patient/5 years
19 Mayo Clinic, Cancer. 201120 Hong and Loughlin, Urology. 2008

- Average amount of diagnosis and monitoring procedures/patient in first 5 years = 14 times Total cost diag/monitoring = (cost of cytology+cystoscopy) X 14 = $479 X 14 = $6,706
- 72,570 new cases/year- Approximately 80% of new cases or 58,056 will survive 5 years= 58,056 X 14 = 812,784 procedures Total costdiag/monitoring = $479 X 812,784 procedures = $389,323,536
Economic ValueEconomic analysis: Current prices

Economic valueEconomic analysis: Pricing/cost estimate
Assay development + Instrumentation Costs$100,000* x 3 proteins = $300,000
(Source: ImmunoChemistry Technologies, LLC.)
Reagent cost per sample:3 proteins X 16 replicates21 = 48 wells/urine sample
4 samples/kit or $156/4 samples = $39/urine sample
ELISA test price to payor: $156 (Source: ImmunoChemistry Technologies, LLC.)
21 Burd, Clin Microbiol Rev. 2010

Projected prices for the diagnosis & monitoring of bladder cancer
$479 + $156 = $635
$156
Price of ELISA kit = $156

Economic valueEconomic analysis: Projected savings
Estimated cost of procedure with biomarker = $156
Initial cost of diagnosis = $479 + $156 = $635Diagnosis and monitoring costs over 5 years = $635 + (13 X $156) = $2663/patient
For 5 year survivors, total cost of monitoring = 58,056 X $2663 = $154,603,228
Therefore, diagnosis and monitoring cost savings = $389,323,536 - $154,603,228= $234,720,308

Market assessmentEconomic analysis: Projected value
Unit demand = Expected number of tests carried out per year = 812,784/5 years = 162,556 tests/yearPrice that payors would pay for test = $156Expected annual revenue = = $156/test X 162,556 tests = $25,358,736
Total Costs = Assay development cost + cost of reagentsAssay development cost = $300,000Cost of reagents = $39 X 162,556 = $6,339,684Total Cost = $6,339,684 + $300,000 = $6,639,684
Potential Profit = $25,358,736 - $6,639,684 =
$19,019,052

Investment Recommendation- High clinical validity and accuracy as a biological indicator- Non-invasive and easy long-term monitoring- Cost-effective- High unit demand
Based on our opportunity analysis, we would strongly recommend investing in this biomarker


APPENDIX

Signs and symptoms
- Hematuria (blood in urine)- Urgent need to empty bladder- Frequent urination- Needing to strain (bear down) when urinating- Pain during urination
Source: National Cancer Institute, What You Need to Know About Bladder Cancer,

Bladder anatomy-Inner layer: Inner lining of bladder. Composed of transitional cells, 7 cell layers thick. Capable of stretching to accommodate urine.
-Middle layer: Smooth muscle. Contraction of muscle squeezes urine out during urination.
- Outer layer: External layer containing fat, fibrous tissue and blood vessels.

Transitional Cell Carcinoma- 90% of bladder cancer occurs in transitional cells (NCI)
- Known as transitional cell carcinoma (TCC) or urothelial cell carcinoma (UCC). (World Health Organization)

Potential BiomarkersPotential biomarkers:1) Protein assays2) miRNA3) DD23 monoclonal antibody4) YAP-15) Metallothionein6) Prefoldin

IL8 (Interleukin-8) & Cancer- A chemokine (signalling factor) produced by Endothelial cells and macrophages. “Many studies have shown overexpression of IL-8 by tumor cells, often induced in response to chemotherapeutic interventions or environmental stresses such as hypoxia.”“The induction of IL-8 signaling activates multiple upstream signaling pathways that (a) impinge on gene expression via regulation of numerous transcription factor activities, (b) modulate the cellular proteome at the level of translation, and/or (c) effect the organization of the cell cytoskeleton through posttranslational regulation of regulatory proteins. As a consequence of the diversity of effectors and downstream targets, IL-8 signaling promotes angiogenic responses in endothelial cells, increases proliferation and survival of endothelial and cancer cells, and potentiates the migration of cancer cells, endothelial cells, and infiltrating neutrophils at the tumor site. Accordingly, IL-8 expression correlates with the angiogenesis, tumorigenicity, and metastasis of tumors in numerous xenograft and orthotopic in vivo models. ”
Waugh DJ, Wilson C. Clin Cancer Res. 2008 Nov 1;14(21)

IL8 (Interleukin-8) & Cancer- A chemokine (signalling factor) produced by Endothelial cells and macrophages. “Many studies have shown overexpression of IL-8 by tumor cells, often induced in response to chemotherapeutic interventions or environmental stresses such as hypoxia.”“The induction of IL-8 signaling activates multiple upstream signaling pathways that (a) impinge on gene expression via regulation of numerous transcription factor activities, (b) modulate the cellular proteome at the level of translation, and/or (c) effect the organization of the cell cytoskeleton through posttranslational regulation of regulatory proteins. As a consequence of the diversity of effectors and downstream targets, IL-8 signaling promotes angiogenic responses in endothelial cells, increases proliferation and survival of endothelial and cancer cells, and potentiates the migration of cancer cells, endothelial cells, and infiltrating neutrophils at the tumor site. Accordingly, IL-8 expression correlates with the angiogenesis, tumorigenicity, and metastasis of tumors in numerous xenograft and orthotopic in vivo models. ”
Waugh DJ, Wilson C. Clin Cancer Res. 2008 Nov 1;14(21)

APOE (Apolipoprotein E) & Cancer-APOE : “ class of apolipoprotein found in the chylomicron and Intermediate-density lipoprotein (IDLs) that is essential for the normal catabolism of triglyceride-rich lipoprotein constituents.[1]” http://en.wikipedia.org/wiki/Apolipoprotein_E“TAF secreted TGFβ induces TNFα expression in breast CSCs. TNFα binds TNFR1 on breast CSCs and activates the PPARα/HIF1α interplay which up-regulates miR130b expression. The interplay is counterbalanced by PPARγ via miR17-5p up-regulation. In turn, the PPARα/HIF1α interplay regulates CAIX, ApoE, IL6 and SLUG expression.”
Source: Papi et al PLoS One. 2013;8(1):e54968

VEGF (Vascular Endothelial Growth Factor) & CancerVEGF: Vascular endothelial growth factor, a signalling protien that induces angiogenesis.VEGF is upregulated during hypoxia.
Source: Vishnu et al, Onco Targets Ther. 2011; 4: 97–113.

Role of APOE , IL8, VEGF in bladder cancer
Source: Waugh DJ, Wilson C. Clin Cancer Res. 2008 Nov 1;14(21)

Biomarker Selection- We selected the 3-protein (VEGF, ApoE and IL8) assay as a biomarker.
- Advantages over current protocol:--

Performance of urinary biomarker combinations
Source: Goodison S, Chang M, Dai Y, Urquidi V, Rosser CJ. PLoS One. 2012;7(10)

Cost of cystoscopy + cytology = $11846/cancer (Kamat et al., 2010)Mean number of subsequent tumor episodes = 3.8 (Soloway et al., 2003)Total cost of initial assessment and surveillance/maintenance =$11846/cancer X 1 initial cancer = $11846$11846/cancer X 3.8 subsequent cancer episodes = $45014.80= $56860.80

3 Protein marker - Pricing assessment1. Source: BDBiosciencesIL-8 kit. (~$490/kit)
2. Refer to assay development doc. Cheaper price/kit (~$156/kit)
3. 2 x 96-well plates/kit. How many replicates for each patient sample for each protein? 2 to 4 replicates (Source: Burd, Clin Microbiol Rev. 2010 July; 23(3): 550–576. CLSI/NCCLS. 2003. Evaluation of the linearity of quantitative measurement procedures: a statistical approach.Approved guideline. CLSI document EP6-A. Clinical
and Laboratory Standards Institute, Wayne, PA.)









![Research Paper Establishing the ... - Journal of Cancer · bladder cancer (NMIBC) [3]. Currently, despite the progress in diagnostic techniques and the improvement in surgical and](https://static.fdocuments.us/doc/165x107/5f4d97bf68593756d475e762/research-paper-establishing-the-journal-of-cancer-bladder-cancer-nmibc-3.jpg)



![Establishing the prediction models for ... - Journal of Cancer · non-muscle-invasive bladder cancer (NMIBC) [3]. Currently, despite the progress in diagnostic techniques and the](https://static.fdocuments.us/doc/165x107/5f4d97bf68593756d475e764/establishing-the-prediction-models-for-journal-of-non-muscle-invasive-bladder.jpg)