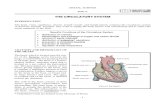
Benzene Flow Chart for Alevel chemistry
-
Upload
syed-umair-anwer -
Category
Documents
-
view
215 -
download
0
Transcript of Benzene Flow Chart for Alevel chemistry

7/23/2019 Benzene Flow Chart for Alevel chemistry
http://slidepdf.com/reader/full/benzene-flow-chart-for-alevel-chemistry 1/1
NO2 NH2 N2+Cl
-OH
Conc nitric & conc
sulphuric
Reflux 55oC
C6H6 + HNO3 C6H5NO2 + H2O
nitrobenzene
Phenyamine
aniline Benzene diazonium chloride phenol
Tin (Sn) & Conc
HCl
Reflux
C6H5NO2 + 6[H] C6H5NH2 + H2O
Nitrous acid and
HCl (use NaNO2)
Below 10oC
C6H5NH2 + HNO2 + HCl C6H5N2+Cl
- + 2H2O
Water
Warm above 10oC
C6H5N2+Cl
- + H2O C6H5OH + N2 + HCl
NO2+
electrophile
CH3+
electrophile
Chloromethane and
anhydrous aluminium
chloride AlCl3
room temp
dry inert solvent
(ether)
CH3
CH3C+
Oelectrophile
Acyl chloride and
anhydrous aluminium
chloride AlCl3
room temp
dry inert solvent
(ether)
O
C - CH3
CH3 CH2Cl
Cl2 and anhydrous
aluminium chloride Fe
or FeCl3 or AlCl3
reflux
Cl
Cl2 and hv
Reflux with KMnO4
O
C - H
O
C - OH
+ NaOH C6H6O- Na+ + H2O
+ Na C6H6O- Na+ + H2
+ Carbonate no reaction
Slightly acidic
Reflux with KMnO4
Benzaldehyde Benzoic Acid
Enhanced reactivity with
aqueous Br2 when compared
with benzenedue to OH directing (No Catalyst
Required)
White ppt
OH
Br
BrBr
Enhanced reactivity with
aqueous HNO3 when compared
with benzene
due to OH directing
mixture of 2 and 4 nitrophenol
OH
NO2
24
3
NH2
Br
BrBr
Enhanced reactivity with
aqueous Br2 when comparedwith benzene
due to NH2 directing
White ppt
Electrophylic
Substitution
OH N=N-
Azo group, stepping stone for delocalized electrons
+
NOT Carboxylic acid unless NaOH
BUT acyl chloride
Ester
NaOH Catalyst
CH2 - OH
Benzyl Alcohol
(Phenyl methanol)
Chlorine and anhydrous
aluminium chloride AlCl3
or Fe or FeCl3
reflux
dry inert solvent (ether)
Cl
5
6
Phenylethanone
O
C – O-
O
C - CH3
iodoform
NaOH and I2
+ CHI3 yellow ppt
O
O
CH3-C-
Phenoxide
CH(OH)CH3
Benzyl Alcohol
(Phenylethanol)
Nucleophilic substitution only under extreme
conditions due to enhanced polarity of C-Cl
compared to C-H bond in benzene
(Conditions NaOHaq 300oC and 300atm)
OH
More acidic than
phenol due to
halogen
Methylbenzene
Toluene
C-Cl bond ispolar due tochlorines electronegative.In thiscase,this isoffset to someextent by the
movement of electrons back towardsthering in the delocalisation.
http://www.chemguide.co.uk/organicprops/arylhalides/background.html#top