Agenda: Warm-up: Explain the solvation process Review – terms, importance, “likes dissolve...
-
Upload
elvin-walton -
Category
Documents
-
view
216 -
download
0
Transcript of Agenda: Warm-up: Explain the solvation process Review – terms, importance, “likes dissolve...
Agenda: Warm-up: Explain the solvation process
Review – terms, importance, “likes dissolve likes”
Solubility Curves What can they tell us about solutions?
Measuring concentration of solutions
Homework: New Element Project
Warm-up: Solvation ProcessExplain what is happening when an ionic
compound is dissolved in water (called solvation or hydration)
View the video - what are key steps that need?How does the water molecules interact with
the ionic compound?
http://group.chem.iastate.edu/Greenbowe/sections/projectfolder/flashfiles/thermochem/solutionSalt.html
a. Making Solutions: Molecules (Covalent compounds)
General Rule: “Likes dissolve likes”
Polar solutes can dissolve in polar solvents.Examples:
Organic solventsNon-polar solute can dissolve in non-polar solvents
Examples: To remove oily stains in dry-cleaning
Lipids will dissolve in hydrocarbonTo clean oil based paintsTo manufacture plastics, man-made
fibers, adhesives
Solution processAnimations https://
www.youtube.com/watch?v=EBfGcTAJF4o
b. Making Solutions: Factors that impact the solubility of solids in liquids
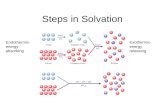
Energy is involved – 2 stepsEndothermic to separate solute & solvent particlesExothermics – attraction between solvent & solute
particlesFactors affecting solids dissolving in liquids
General trend to increase solubility
Chemical components of solute and solvent
Intermolecular forces High attraction between solute and solvent particles
Temperature
Particle size
Agitation (amount of stirring)
c. Measuring ConcentrationSolubility: number of grams of solute in 100
grams of water at 20℃ Solubility Curves
Saturated solutionsUnsaturatedSupersaturated
http://www.youtube.com/watch?v=XSGvy2FPfCw http://www.youtube.com/watch?
v=1y3bKIOkcmk&feature=related
Interpreting solubility curvesY axis
X axis
Lines
If the amount needed is more or less than 100 g of H₂O
1 g H₂0 = 1 mL H₂0
c. Making solutions: Factors that affect the solubility of gases in liquids
Factors affecting gases
General trends to increase solubility
Temperature
Pressure
Think about soda (carbonated drinks)
Category Ions Except with these ions
Examples
Soluble cations
Group 1 ions and ammonium, NH4
+No exceptions
Na2CO3, LiOH, and (NH4)2S are soluble.
Soluble anions
NO3- and
C2H3O2- No exceptions
Bi(NO3)3, and Co(C2H3O2)2 are soluble.
Usually soluble anions
Cl-, Br-, and I-
Soluble:Exceptions: Ag+ and Pb2+
CuCl2 is water soluble, but AgCl is insoluble.
SO42-
Soluble Exceptions:Ba2+ and Pb2+
FeSO4 is water soluble, but BaSO4 is insoluble.
Usually insoluble anions
CO32-, PO4
3-, and OH-
Insoluble: Exceptions: group 1 elements and NH4
+
CaCO3, Ca3(PO4)2, and Mn(OH)2 are insoluble in water, but (NH4)2CO3, Li3PO4, and CsOH are soluble.
Solubility Rules determined under specified concentration (often 0.1% molarity)
d. Measuring Concentrations Molarity (M) by definition =
Molarity = Moles of solute 1 Liter of solution
Therefore: 2 molar solution = __________________
Discovery Education video Standard Deviants School Chemistry: solutions &
dilutions
Molarity problems M= moles_______ 1 Liter solutionWhat I s the molarity of a solution in which
58 gram of NaCl are dissolved in 1.0 L of solution?
What is the molarity of a solution in which 10.0 grams of silver I nitrate is dissolved in 500 mL of solution?
How many grams of potassium nitrate should be used to prepare a 2.0 L of a 0.5 molar solution?
Steps needed in molarity calculationsMoles
For grams:
Liters of solutionIf less than 1L ? mL = 1L
Comparison __________ = moles of solute1 liter of solution
Molarity practiceTo what volume should 5.0 g of KCl be diluted
in order to prepare a 0.25M solution?
How many grams of copper II sulfate – penta hydrate are needed to prepare 100 mL of a 0.10M solution?
9g. Dilution of concentrated solutions: V₁M₁= V₂M₂Molarity by dilution practice problems
Most reagents are sold & sorted in concentrated solutions
How much concentrated 18M sulfuric acid is needed to prepare 250mL of a 6.0M solution?
How much concentrated 12M hydrochloric acid is needed to prepare 100 mL of a 2.0 M solution?
To what volume should 25 mL of 15 M nitric actid be diluted to prepare a 3.0 M solution?
To how much water should 50 mL of 12 M hydrochloric acid be added to produce a 4.0 M solution?
To how much water should 100 mL of 18M sulfuric acid be added to prepare 1.5 M solution?
Colligative PropertiesThe properties of the solution that depend on the
number of particles in solution, not the identity of the solute. The solvent properties will be changed.
NaClCaCl₂AlCl₃
Melting point depression; boiling point elevation; Vapor pressure lowering
Resourceshttp://
www.karentimberlake.com/solution.htm
http://www.afn.org/~afn02809/powerpoint-list.htm
http://www.chemistrygeek.com/chemistrypowerpoint/Student%20Ch%2015%20Solutions.ppt
http://college.cengage.com/chemistry/general/zumdahl/world_of_chem/1e/instructors/ppt/figures/viewindex.html