6a Amino Acids
-
Upload
kamal-jaoni -
Category
Documents
-
view
214 -
download
0
Transcript of 6a Amino Acids
-
8/2/2019 6a Amino Acids
1/4
1
Amino Acid
SEPARATION OF AMINO ACIDS BYSEPARATION OF AMINO ACIDS BYSEPARATION OF AMINO ACIDS BYSEPARATION OF AMINO ACIDS BYSEPARATION OF AMINO ACIDS BYTHIN LAYER CHROMATOGRAPHYTHIN LAYER CHROMATOGRAPHYTHIN LAYER CHROMATOGRAPHYTHIN LAYER CHROMATOGRAPHYTHIN LAYER CHROMATOGRAPHY
MMMMMATERIALSATERIALSATERIALSATERIALSATERIALS NNNNNEEDEDEEDEDEEDEDEEDEDEEDED
silica gel plate mobile phase: 1-butanol, glacial
acetic acid and water (4:1:1) known solutions of amino acids unknown solutions ofamino acids micropipette developing tank 2% ninhydrin solution
heat gun pencil gloves
PURPOSE:PURPOSE:PURPOSE:PURPOSE:PURPOSE: To understand the concepts of chromatography and toidentify unknown amino acids.
CAUTION:CAUTION:CAUTION:CAUTION:CAUTION: 1) Refrain from prolonged exposure to the mobile phasesolvent (1-butanol, glacial acetic acid and water). Keep thecover on the developing tank when not adding or removing aplate. The 1-butanol will evaporate. 2) Ninhydrin will stainyour skin. Wear gloves to protect yourself from contaminationand from contaminating your silica gel plate.
BACKGROUNDBACKGROUNDBACKGROUNDBACKGROUNDBACKGROUND
The discovery ofchromatography in 1944revolutionized theseparation and detection ofamino acids and dipeptides.The separation is based onthe liquid-liquid partitionof the compounds between
two immiscible phases.Initially the separationswere primarily conducted onfilter paper and werecalled paper chromatography.
In paper chromatography the hydrated cellulose fibers of thepaper act as the stationary phase. A polar solvent ascends inthe vertically held paper by capillary action and is the mobilephase. In thin layer chromatography (TLC) a thin uniform layerof silica gel acts as the stationary phase. TLC is replacing
paper chromatography because the plates are easier to use thanthe paper, they give a sharper separation and the amino acids ordipeptides can easily be collected from the plate.
Many microscopic distributions of the compounds occur betweenthe mobile and the stationary phases. In time equilibrium isestablished between the two phases and the more solublecompounds move farther along the plate; different compounds move
-
8/2/2019 6a Amino Acids
2/4
2
Amino Acids
different distances from the origin. The plate is dried, sprayedwith a ninhydrin solution and heated in order to locate theamino acids. The ninhydrin reacts with the amino acids to formcolored products.
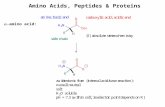
The ratio of the distance moved by the amino acids to thedistance moved by the solvent front from the original spot onthe paper is defined as the R
fvalue and is characteristic of the
compound.
RRRRRfffffcompound =compound =compound =compound =compound = distance traveled by compounddistance traveled by compounddistance traveled by compounddistance traveled by compounddistance traveled by compound
distance traveled by solventdistance traveled by solventdistance traveled by solventdistance traveled by solventdistance traveled by solvent
Rfvalues depend on several factors: type of silica gel plus
binder used, water content of the thin layer, concentration ofsolute, temperature, manner of development and distance of the
starting point from the solvent. Known compounds are usually runon the same plate as the unknowns to assist in identification ofthe unknowns rather than relying solely on published R
fvalues.
PROCEDUREPROCEDUREPROCEDUREPROCEDUREPROCEDURE
1. Put gloves on. If your developing tank isn't prepared, addenough solvent to a depth of approximately 1 cm or less.
2. Get your silica gel plate. Carefully hold the plate by the
sides to prevent disturbing the silica gel layer. Draw apencil line about 1.5 cm from the bottom plate.
3. Mark one point onthe line for each oneof your known andunknown solutions. (Ifyou have four knownsolutions and 2unknowns, mark sixpoints.) Leave margins
of at least 1.5 cm onboth sides. Number eachpoint.
4. At point number 1apply a very small dropof one of your knownsolutions. Do not wet1 2 3 4 5 6 7
-
8/2/2019 6a Amino Acids
3/4
3
Amino Acid
the silica beyond a diameter of 2-3 mm. Locating the centerof large spots will be difficult later when the spot hasmoved along the paper.
5. After the liquid has evaporated (only a few seconds), add a
second drop to the same spot. Record the name of the aminoacid and the number of the spot.
6. Repeat this procedure for the remaining solution. Remember torecord the name of the amino acid or unknown number and thenumber of the spot.
7. Allow all the spots to dry completely.
8. Place your TLC plate in the developing tank with the mobilephase with the spots toward the bottom.
9. Allow the solvent to ascend the silica gel to at least 3/4of its height, which will to require 1 hour or less. (Thefarther the solvent ascends, the greater the separation.Immediately remove the plate, if the solvent reaches thetop.)
10. Remove the plate and quickly mark the farthest advance ofsolvent front with a pencil, unless it reached the top ofthe paper.
11. Dry the plate with a heat gun. Be careful to move the heatgun around and not heat one point continuously. Do thisprocedure in the hood.
12. Spray the plate with the 2% ninhydrin solution in the hood.
13. Do not allow the ninhydrin solution to stream down theplate, because this may move some of the compounds.
14. Dry the plate again with the heat gun. Do not over heat the
plate. Long heating times may cause browning of the plateover the entire surface.
15. Circle each colored spot with a pencil. The ninhydrin spotsfade gradually, so circle at once.
16. Measure the distance from the origin to the center of eachcolored spot and calculate the R
fvalues for all spots.
-
8/2/2019 6a Amino Acids
4/4
4
Amino Acids
17. Record the Rfvalues and the color of
each ninhydrin spot.
18. Identify the unknown amino acids.
QUESTIONSQUESTIONSQUESTIONSQUESTIONSQUESTIONS
1. Why does touching the silica gel withyour hands potentially contaminateyour plate?
2. Why can an Rfvalue never be greater
than 1?
3. What would happen if so much solventwas used (mobile phase) that the
original spots were covered withsolvent?
4. What would happen if you made theline and points with an ink penrather than a pencil?
5. You dropped and mixed up yoursamples. You know that one containsonly valine, one contains valine and
glycylvaline, one contains valine and alanine, one containsonly glycine and valine, and one contains glycine andglycylleucine. How would you determine what your samples areusing TLC and the data below? Can you figure out what theyall are?
RRRRRfffffValues for Amino Acids and DipeptidesValues for Amino Acids and DipeptidesValues for Amino Acids and DipeptidesValues for Amino Acids and DipeptidesValues for Amino Acids and Dipeptides
CompoundCompoundCompoundCompoundCompound RRRRRfffff
ColorColorColorColorColor
glycine 0.26-0.29 purplealanine 0.39-0.42 purpleglycylvaline 0.62-0.66 grayvaline 0.62-0.64 purpleglycylleucine 0.76-0.80 light brown
Rfvalues taken two days after solvents were mixed and with solvent advance
100 mm in 43 minutes at a temperature of 31 C.
originalline
ninhydrinspot
solventfront
numerator
denom
inator
Calculating RCalculating RCalculating RCalculating RCalculating RfffffValuesValuesValuesValuesValues