2.1 ELEMENTS. The smallest building blocks of matter are atoms. What is the difference between an...
-
Upload
theodore-ross -
Category
Documents
-
view
214 -
download
1
Transcript of 2.1 ELEMENTS. The smallest building blocks of matter are atoms. What is the difference between an...

2.1 ELEMENTS

• The smallest building blocks of matter are atoms.
• What is the difference between an atom and an element?

• An element is a pure substance that cannot be broken down or separated into simpler substances.
• An element is a type of atom.

Example:Copper is an element,
An atom of copper is the smallest possible piece of copper you can have, that still has all the properties of copper.

CHEMICAL SYMBOLS
• Each element has a name, but the names are different depending on the language you speak.

• Symbols provide an international language so all chemists around the world can share their discoveries.– If symbol is only one letter capitalized– If symbol is 2 letters 1st capitalized, 2nd lower
case• Eg: Oxygen = O Mercury = Hg

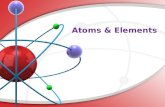
INVESTIGATING THE STRUCTURE OF THE ATOM
• Every atom is composed of 3 types of particles:– Protons: positively charged– Neutrons: no charge– Electrons: negatively charged
• Protons and neutrons are found in the nucleus• Electrons circle the nucleus in particular
energy levels

Rules for atomic particles:
1. # protons = # electrons2. Atomic number = # protons3. Atomic mass = the mass of an average atom
of an element4. Ion charge = an electric charge that forms on
an ion when it gains or loses electrons.

Assignment:
• “Learning chemical symbols” worksheet• Reading check page 47• CYU page 51 #1-18• “Adopt-an-element” project

Who created the periodic table?
• Dmitri Mendeleev• He arranged it by grouping elements with
similar properties together• The periodic table contains an amazing
amount of information

Info on a square in the Periodic Table:
• Some elements have multiple ion charges as they form ions in more than one way.

Trends in the periodic table:
• Elements are arranged in order of atomic number
• THEY ARE NOT ARRANGED BY ATOMIC MASS!!

Trends in the periodic table:
• Elements on the LEFT side of the table mostly form POSITIVE ions– This means they LOSE ELECTRONS


Trends in the periodic table:
• Elements on the RIGHT side of the table mostly form NEGATIVE ions– This means they GAIN ELECTRONS


Trends in the periodic table:• Elements
in the same column usually have similar ion charges.

Trends in the periodic table:
Metals, Non-metals and Metalloids…
• Elements are clumped in groups that have similar properties
• Table 2.2 (pg.55) shows the properties that each of these groups have in common.

Trends in the periodic table:• Within each of these groupings are columns of
elements with very similar properties. – These are called families

Assignment:
• Reading Check page 57• 2.2 Worksheet• Complete “Adopt an Element” research