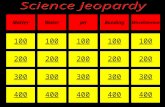
100 200 400 300 400 OrangeGreenGreyBlue 300 200 400 200 100 500 100.
-
Upload
donald-stewart -
Category
Documents
-
view
238 -
download
0
Transcript of 100 200 400 300 400 OrangeGreenGreyBlue 300 200 400 200 100 500 100.


100 100
200 200
400 400
300
400
Orange Green Grey Blue
300 300 300
200
400
200
100
500 500 500 500
100

Ionic bond
when an atom lends one or more electrons to another that receives them.

Covalent bond
Bond that forms when atoms share electrons

Atomic number
The periodic table of the elements arranges the elements in order of increasing …..

metals
Tend to be hard, shiny, and are good conductors of heat and electricity.

Non-metals
tend to have dull surfaces, are good insulators but poor conductors of heat and electricity

periods
the horizontal rows on a periodic table

Families
the vertical columns on a periodic table

Noble gases
the most stable elements on the periodic table of the elements.

liquids
Mercury and bromine exist as ________ at room temperature

gases
Hydrogen and oxygen exist as gases at room temperature

solids
Copper and lead exist as ______ at room temperature.

Physical
Length, mass, volume, and density are examples of _________ properties

chemical
Flammability and corrosiveness are examples of _________ properties

physcial
Tearing a sheet of paper in half is an example of a __________ change

chemical
Burning a sheet of paper is an example of a ______ change

synthesis
A chemical equation in the form of: A + B AB, is an example of a ______ reaction..

decomposition
A chemical equation in the form of: AB A + B, is an example of a ________ reaction.

What technique would you use to find the volume ofAn irregularly shaped rock?
Water displacement

What could you use to separate magnetic iron particles and
non-magnetic sand?
A magnet

8
Oxygen’s atomic number is 8 and its mass is 16, how many protons does it have?